Confirmed Pureness & Third-party Screening
페이지 정보

본문
Artificial SPPS production, managed procedures, and research-grade handling minimize contamination risk. Select an item to watch the most recent COA for the current batch. Neither the statements neither the products of this company objective to detect, deal with, treat, or fend off any kind of disease.
An extensive overview for site - click the next website page, scientists and companies on exactly how to source peptide powders, focusing on quality, purity, and ... Lots of CoA documents are readily available on the product's webpage (click over here now); check for the paper tab in the product's webpage. After completing the information, we will be able to provide the CoA as a pdf. By focusing on the honesty of our study peptides, click the next website page) we make it possible for scientists to concentrate on their work, knowing they have reputable data at their fingertips.
Our dedication to third-party screening is designed to support responsible science-- not unproven or off-label usage. A fairly reduced peptide material can be anticipated when a peptide consists of a big proportion of basic amino acids, specifically arginines. Understanding a COA encourages researchers to seriously examine the products they use. At VELA Peptides, all batches are evaluated and verified with COAs released within six months of sale, making sure transparency and accuracy throughout our brochure. Some suppliers also make use of QR codes on vials that connect straight to the COA for that certain batch. Not "in-house screening" or "tested by our team." An actual independent lab with a name, address, and call details you can validate.
If identity testing is missing out on, you have no evidence the item is what the label states. Some suppliers skip testing totally and just make up documents. Others use real COAs from one batch and slap them on every little thing they market. A Certificate of Analysis only mirrors the material that was examined. Without proper upstream controls and documentation, Can You Take Excessive Collagen In A Day? Specialists Answer a COA alone can not guarantee that succeeding batches match the exact same standard.
Advanced HPLC systems interfaced with chromatography management software program and verified by licensed professionals show severe investment in quality assurance infrastructure. This tools is essential for dependable peptide synthesis and production. Seek labs with ISO/IEC certification or comparable high quality qualifications that demonstrate validated logical techniques. High-Performance Fluid Chromatography divides the target peptide from peptide-related contaminations like deletion series, truncated fragments, and synthesis by-products. The method gauges what portion of the example is your wanted peptide versus other organic impurities.
Certificates of Evaluation (COA) are necessary documents in the world of research study chemicals, cannabinoids, and different other industries, which play a substantial duty in promoting customer safety and mouse click the next article security. Unlike items intended for human usage, research chemicals often call for specific testing procedures to confirm their chemical properties, security levels, and effectiveness. Whether you're a scientist or a maker, comprehending just how to check out a COA can encourage you to make educated decisions about the items you're evaluating, acquiring, or creating. Firstly, at Combination Peptide, we concentrate on supplying high-grade research peptides.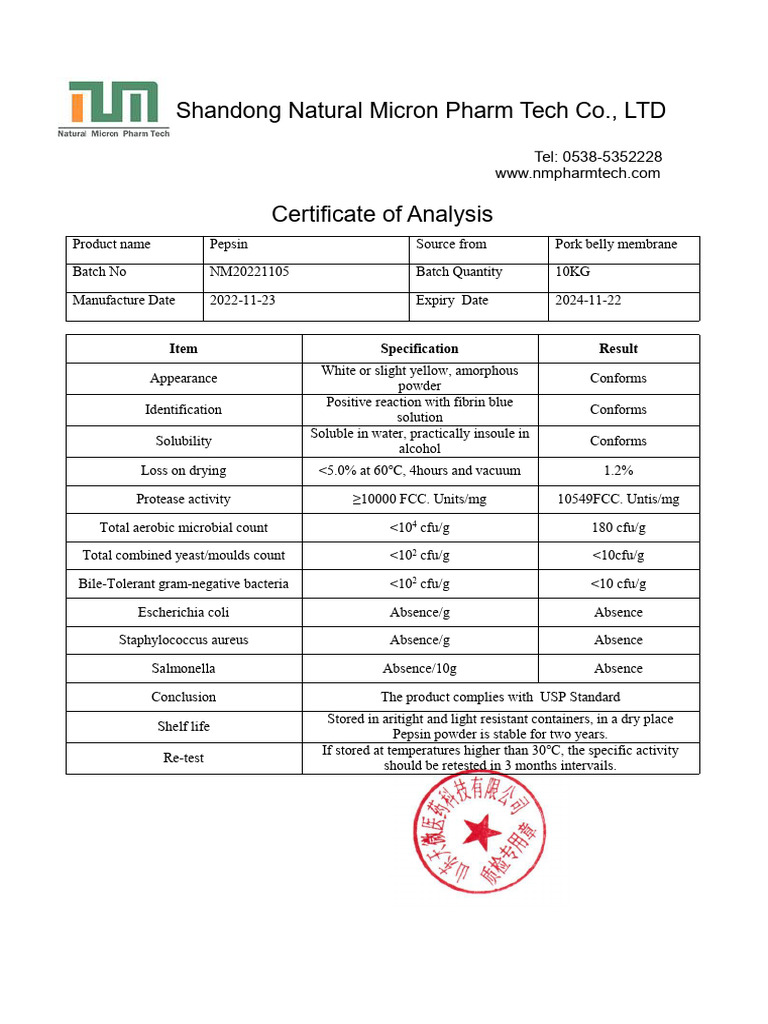

How To Verify Credibility
An extensive overview for site - click the next website page, scientists and companies on exactly how to source peptide powders, focusing on quality, purity, and ... Lots of CoA documents are readily available on the product's webpage (click over here now); check for the paper tab in the product's webpage. After completing the information, we will be able to provide the CoA as a pdf. By focusing on the honesty of our study peptides, click the next website page) we make it possible for scientists to concentrate on their work, knowing they have reputable data at their fingertips.
- Reputable distributors run as authorized services with verifiable identifications and clear call info.
- Basic material costs, synthesis expenditures, and logical screening create standard rates that reputable suppliers can't undercut without giving up top quality.
- A lot of genuine COAs include an URL or QR code that reroutes to the testing research laboratory's main site, where the complete outcomes can be confirmed.
- It's essential to make sure that the COA represents the particular batch or great deal variety of the item you want.
What Is Using A Certification Of Analysis?
What is the cost of a COA Certification?
The COA technique multiple-choice assessment permits candidates to sit for the COA assessment for a preliminary charge of $& #x 24; 150.00. If the prospect passes the assessment, the candidate would certainly pay an additional $& #x 24; 150 to get their certification within thirty days of obtaining their method examination outcomes.
Our dedication to third-party screening is designed to support responsible science-- not unproven or off-label usage. A fairly reduced peptide material can be anticipated when a peptide consists of a big proportion of basic amino acids, specifically arginines. Understanding a COA encourages researchers to seriously examine the products they use. At VELA Peptides, all batches are evaluated and verified with COAs released within six months of sale, making sure transparency and accuracy throughout our brochure. Some suppliers also make use of QR codes on vials that connect straight to the COA for that certain batch. Not "in-house screening" or "tested by our team." An actual independent lab with a name, address, and call details you can validate.
What Should I Do If A Coa Shows Failed Examination Results?
If identity testing is missing out on, you have no evidence the item is what the label states. Some suppliers skip testing totally and just make up documents. Others use real COAs from one batch and slap them on every little thing they market. A Certificate of Analysis only mirrors the material that was examined. Without proper upstream controls and documentation, Can You Take Excessive Collagen In A Day? Specialists Answer a COA alone can not guarantee that succeeding batches match the exact same standard.
Advanced HPLC systems interfaced with chromatography management software program and verified by licensed professionals show severe investment in quality assurance infrastructure. This tools is essential for dependable peptide synthesis and production. Seek labs with ISO/IEC certification or comparable high quality qualifications that demonstrate validated logical techniques. High-Performance Fluid Chromatography divides the target peptide from peptide-related contaminations like deletion series, truncated fragments, and synthesis by-products. The method gauges what portion of the example is your wanted peptide versus other organic impurities.
Certificates of Evaluation (COA) are necessary documents in the world of research study chemicals, cannabinoids, and different other industries, which play a substantial duty in promoting customer safety and mouse click the next article security. Unlike items intended for human usage, research chemicals often call for specific testing procedures to confirm their chemical properties, security levels, and effectiveness. Whether you're a scientist or a maker, comprehending just how to check out a COA can encourage you to make educated decisions about the items you're evaluating, acquiring, or creating. Firstly, at Combination Peptide, we concentrate on supplying high-grade research peptides.

- 이전글한인약국 정품 남성건강 구매 가이드 26.04.02
- 다음글우즐성 비아그라 제품 특징 이용 가이드 , 이용 가이드 안내 26.04.02
댓글목록
등록된 댓글이 없습니다.

